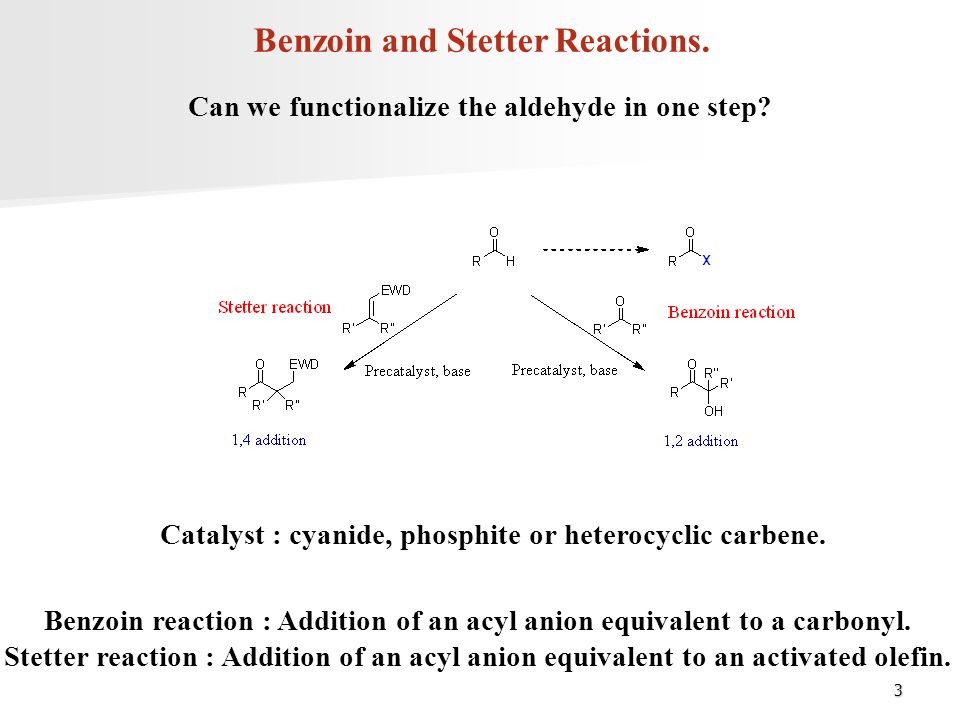
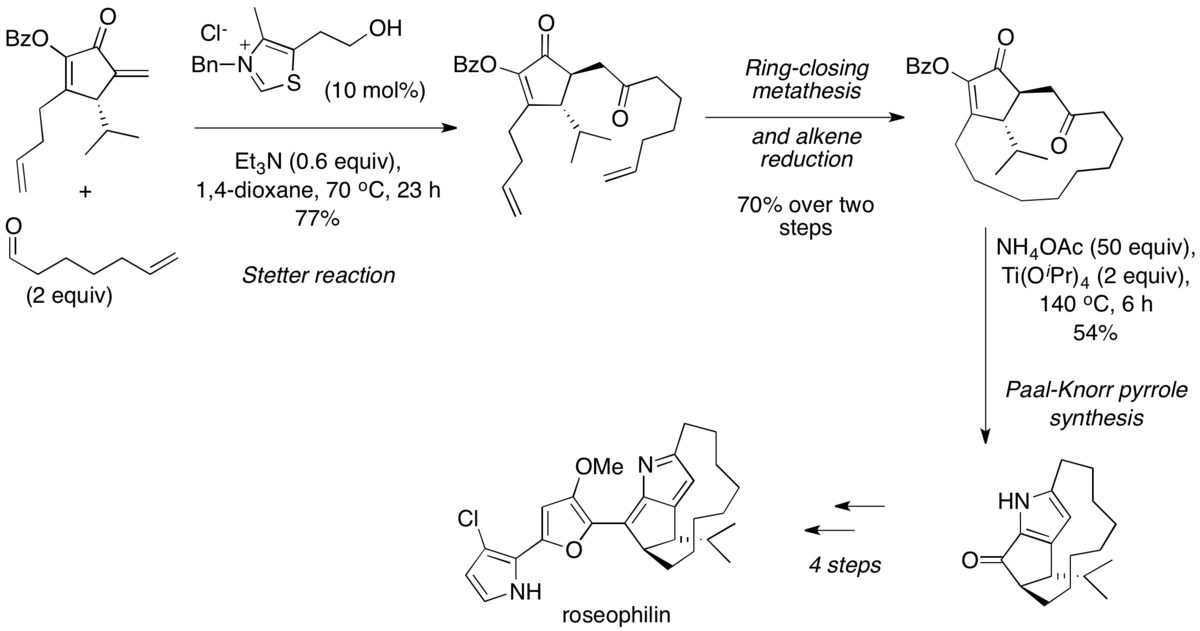
Multicatalytic, asymmetric Michael/Stetter reaction of salicylaldehydes and activated alkynes | PNAS

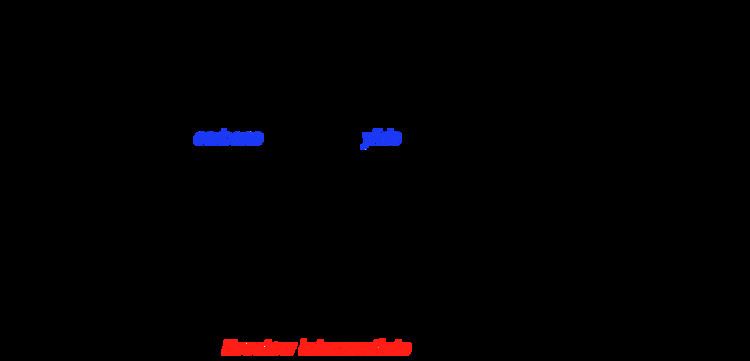
Scheme 3 Proposed reaction pathway for the thiazolium-catalyzed Stetter... | Download Scientific Diagram

Scheme 2 Proposed reaction pathway for the thiazolium-catalyzed Stetter... | Download Scientific Diagram
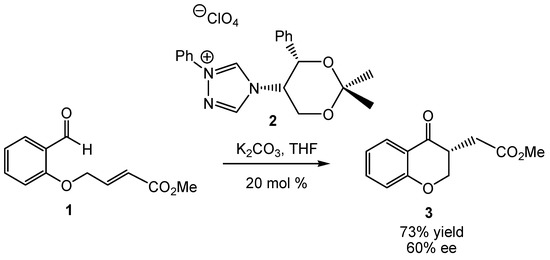
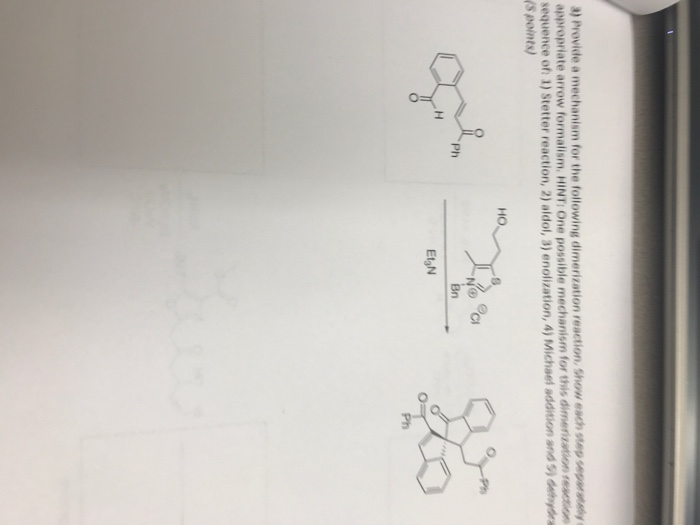
Molecules | Free Full-Text | Understanding the Mechanism of the Intramolecular Stetter Reaction. A DFT Study | HTML

Molecules | Free Full-Text | Understanding the Mechanism of the Intramolecular Stetter Reaction. A DFT Study | HTML

Insights into the Competing Mechanisms and Origin of Enantioselectivity for N-Heterocyclic Carbene-Catalyzed Reaction of Aldehyde with Enamide | Scientific Reports
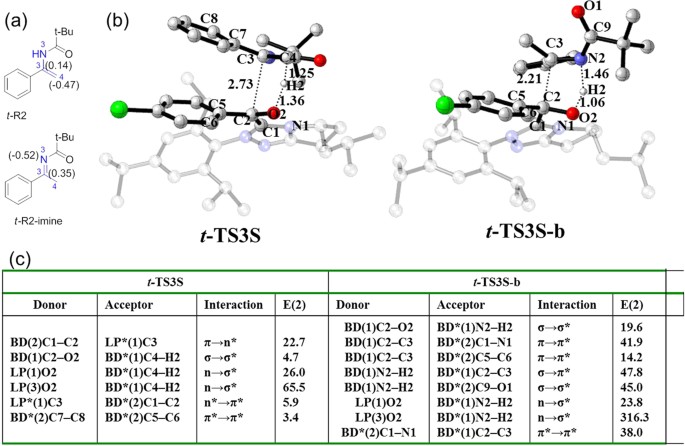
Insights into the Competing Mechanisms and Origin of Enantioselectivity for N-Heterocyclic Carbene-Catalyzed Reaction of Aldehyde with Enamide | Scientific Reports