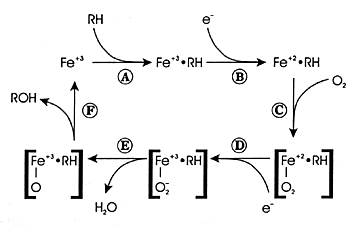
![PDF] Cytochrome P 450 Enzymes in Oxygenation of Prostaglandin Endoperoxides and Arachidonic Acid Cloning , Expression and Catalytic Properties of CYP | Semantic Scholar PDF] Cytochrome P 450 Enzymes in Oxygenation of Prostaglandin Endoperoxides and Arachidonic Acid Cloning , Expression and Catalytic Properties of CYP | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/89998bdde116a1d089b0587a65f5da50f13f536b/8-Figure1-1.png)
PDF] Cytochrome P 450 Enzymes in Oxygenation of Prostaglandin Endoperoxides and Arachidonic Acid Cloning , Expression and Catalytic Properties of CYP | Semantic Scholar
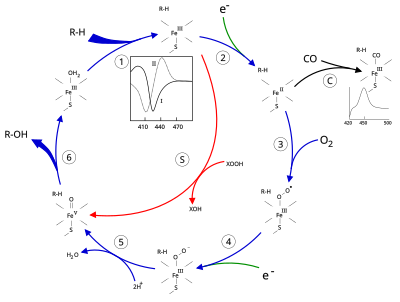
Canonical reaction mechanism for substrate (Sub) hydroxylation by CYPs. | Download Scientific Diagram

The 141st Annual Meeting of the Pharmaceutical Society of Japan (Hiroshima)/Metabolic <em>ipso</em>-Substitution of 5-Substituted-2-oxindole Catalyzed by Cytochrome P450
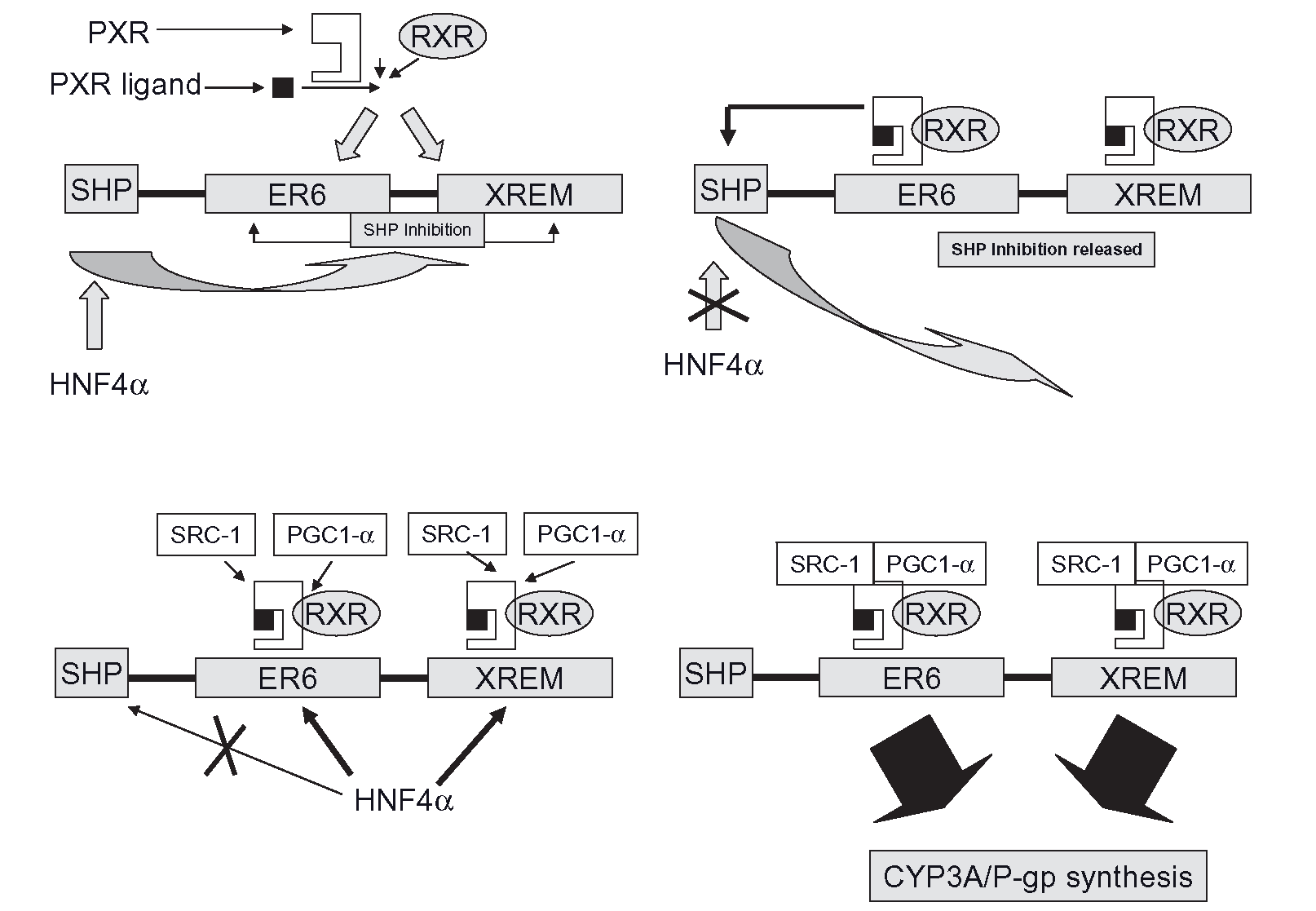
Mechanisms of enzyme induction (Induction of Cytochrome P450 Systems) (Human Drug Metabolism) Part 2
Deuterium isotope effects in drug pharmacokinetics II: Substrate-dependence of the reaction mechanism influences outcome for cytochrome P450 cleared drugs

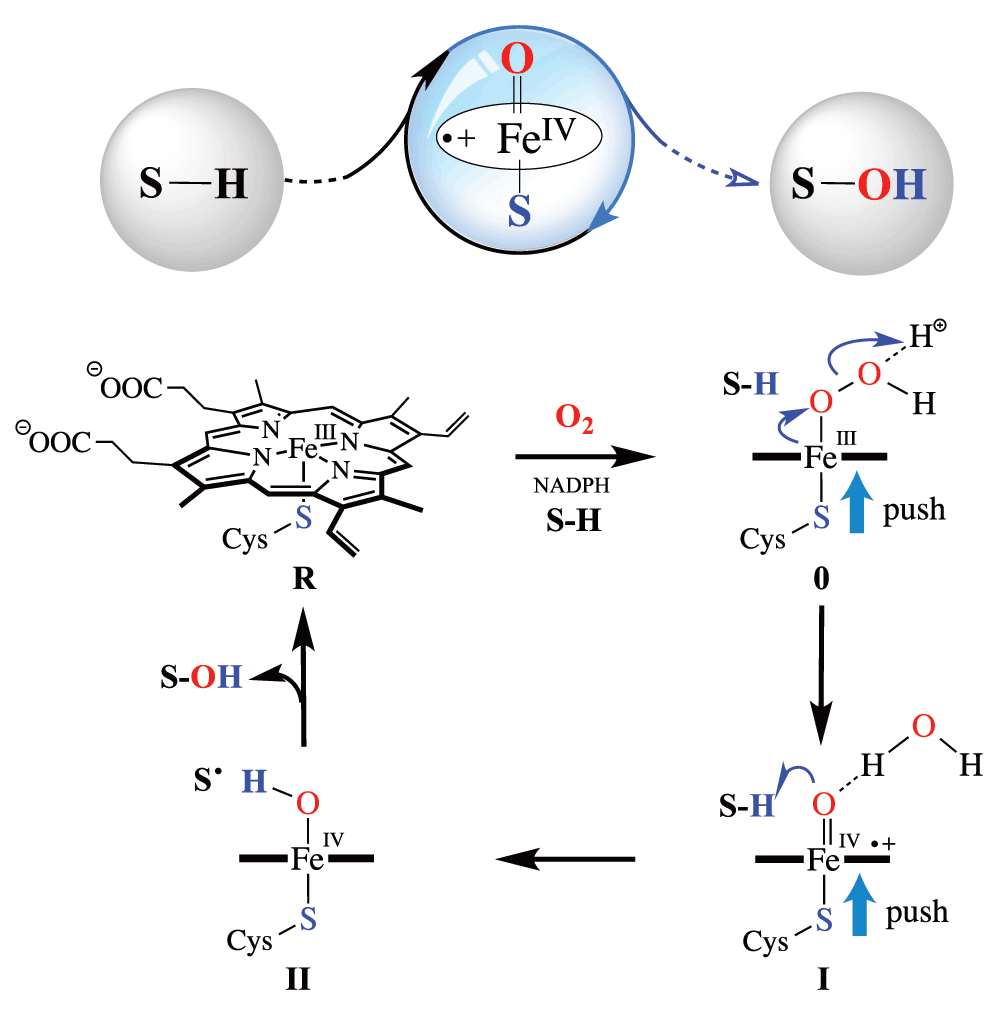
Metabolic‐intermediate complex formation with cytochrome P450: Theoretical studies in elucidating the reaction pathway for the generation of reactive nitroso intermediate - Taxak - 2012 - Journal of Computational Chemistry - Wiley Online Library
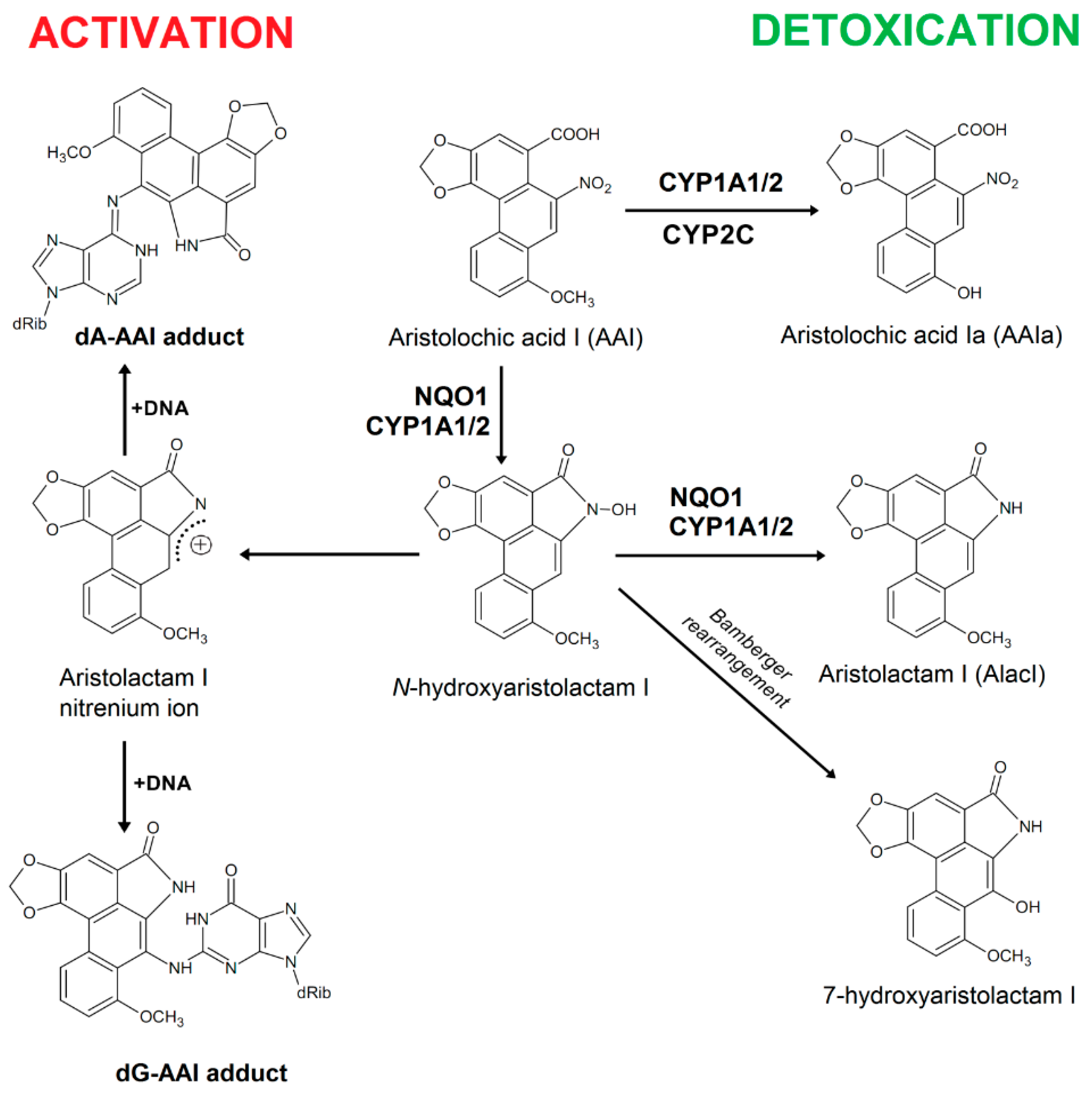
IJMS | Free Full-Text | A Mechanism of O-Demethylation of Aristolochic Acid I by Cytochromes P450 and Their Contributions to This Reaction in Human and Rat Livers: Experimental and Theoretical Approaches

Versatile biocatalysis of fungal cytochrome P450 monooxygenases | Microbial Cell Factories | Full Text