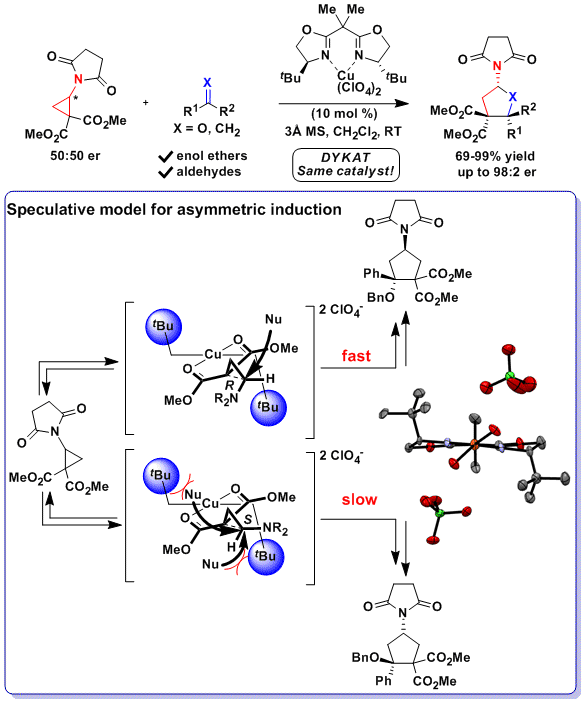
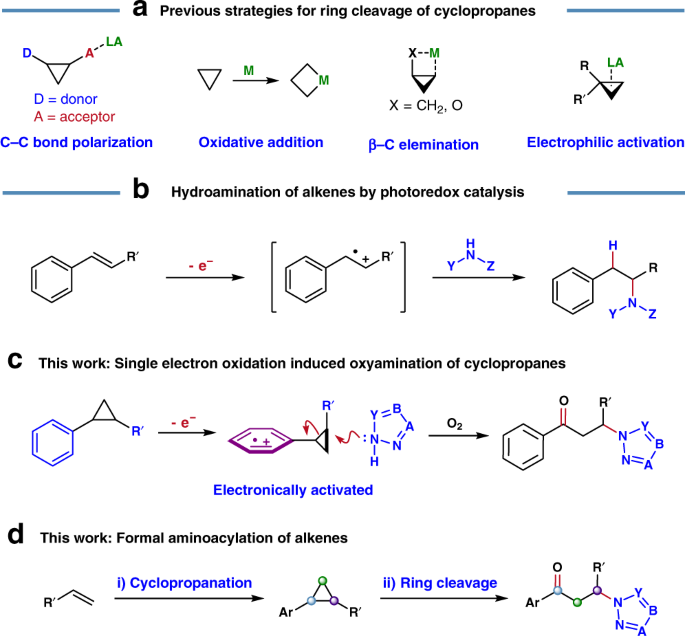
Mild Ring‐Opening 1,3‐Hydroborations of Non‐Activated Cyclopropanes - Wang - 2018 - Angewandte Chemie - Wiley Online Library
Intramolecular donor–acceptor cyclopropane ring-opening cyclizations - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C3CS60238A

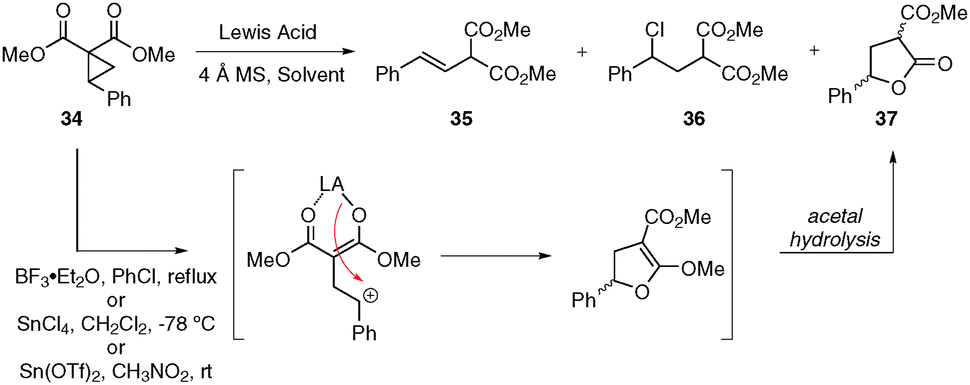
Boron Trifluoride Mediated Ring-Opening Reactions of trans-2-Aryl-3-nitro- cyclopropane-1,1-dicarboxylates. Synthesis of Aroylmethylidene Malonates as Potential Building Blocks for Heterocycles
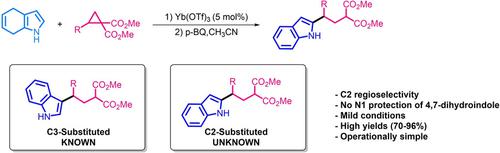
The Reaction of Donor‐Acceptor Cyclopropanes with 4,7‐Dihydroindole: A New Protocol for the Synthesis of Divergent C2‐Alkylated Indoles,ChemistrySelect - X-MOL

Recent Advances in the Chemistry of Doubly Activated Cyclopropanes: Synthesis and Reactivity | Bentham Science
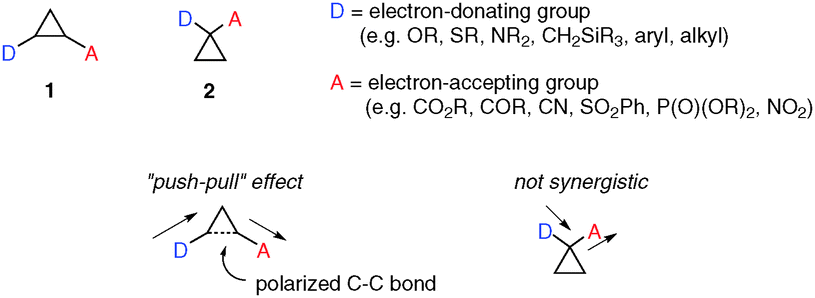
Intramolecular donor–acceptor cyclopropane ring-opening cyclizations - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C3CS60238A

Synthesis and biological evaluation of bromophenol derivatives with cyclopropyl moiety: Ring opening of cyclopropane with monoester - ScienceDirect
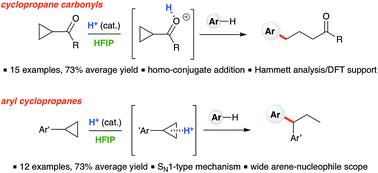
Ring-opening hydroarylation of monosubstituted cyclopropanes enabled by hexafluoroisopropanol - Chemical Science (RSC Publishing)
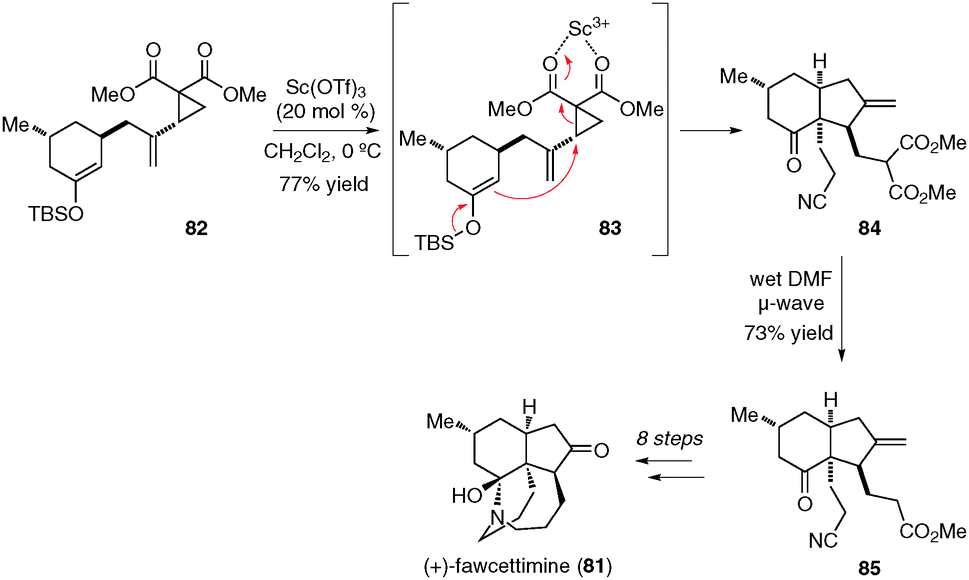
Intramolecular donor–acceptor cyclopropane ring-opening cyclizations - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C3CS60238A
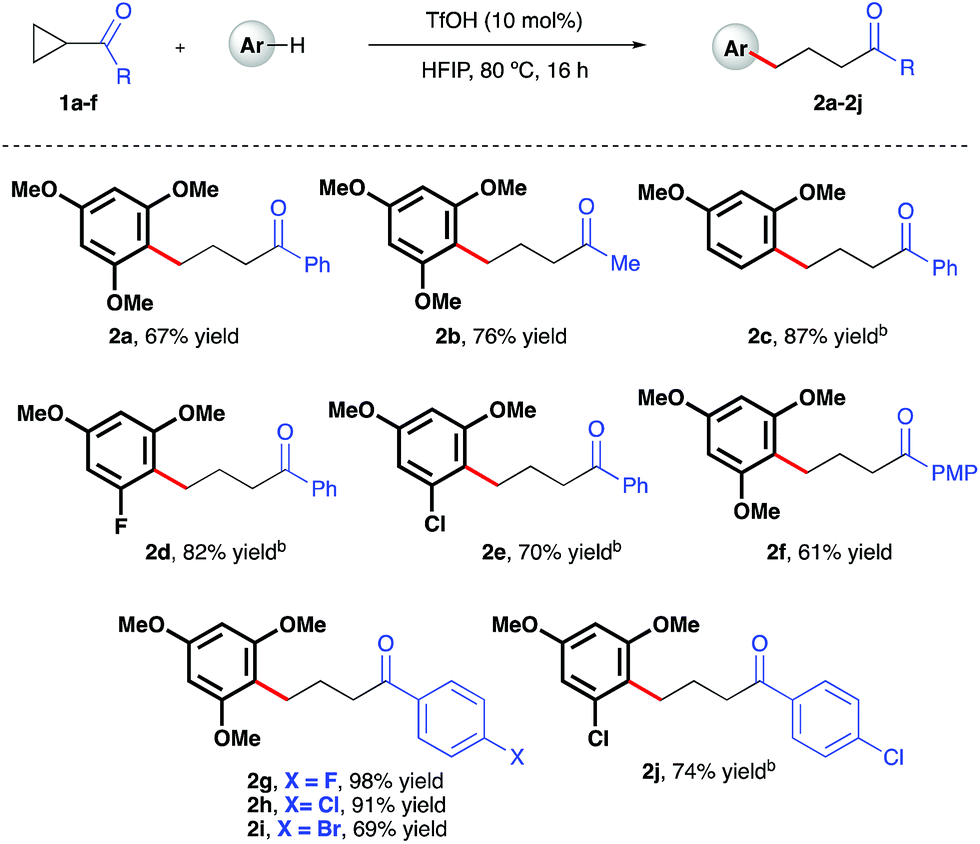
Ring-opening hydroarylation of monosubstituted cyclopropanes enabled by hexafluoroisopropanol - Chemical Science (RSC Publishing) DOI:10.1039/C8SC02126K
![Metal‐Free Ring Opening Cyclization of Cyclopropane Carbaldehydes and N‐Benzyl Anilines: An Eco‐Friendly Access to Functionalized Benzo[b]azepine Derivatives - Dey - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library Metal‐Free Ring Opening Cyclization of Cyclopropane Carbaldehydes and N‐Benzyl Anilines: An Eco‐Friendly Access to Functionalized Benzo[b]azepine Derivatives - Dey - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/843b5375-d568-49d5-9e1f-3a3aeb92b0b6/adsc201801714-toc-0001-m.jpg)





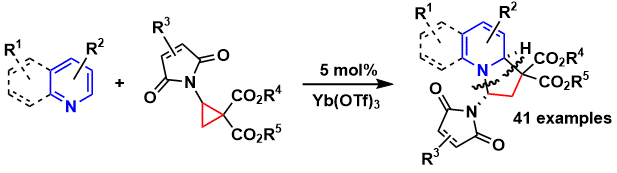


![PDF] Ring Opening of Donor–Acceptor Cyclopropanes with N-Nucleophiles | Semantic Scholar PDF] Ring Opening of Donor–Acceptor Cyclopropanes with N-Nucleophiles | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/9052f234156e8bd52e336c19257b62e75025721a/28-Table3-1.png)